
the unit in which CAPACITANCE is measured. It is usually denoted by the symbol F. For
 example, the capacitance (C) of a conductor measured in farads is the charge (in coulombs) needed to raise the potential by one volt i.e. 1 farad = 1 coulomb per volt (1F = 1 CV-1). As the farad itself is usually too large a quantity for most applications, the practical unit is the microfarad (µF), or one millionth of a farad.
example, the capacitance (C) of a conductor measured in farads is the charge (in coulombs) needed to raise the potential by one volt i.e. 1 farad = 1 coulomb per volt (1F = 1 CV-1). As the farad itself is usually too large a quantity for most applications, the practical unit is the microfarad (µF), or one millionth of a farad.Taken from Dictionary of Science
_____________________________________________________________
The farad (symbol: F) is the SI unit of capacitance. The term farad is named after the English physicist Michael Faraday.
A farad is the charge in coulombs a capacitor will accept for the potential across it to change 1 volt. A coulomb is 1 ampere second. Example: A 47 mA current causes the voltage across a capacitor to increase 1 volt per second. It therefore has a capacitance of 47 mF. It has the base SI representation of s4·A2·m-2·kg-1. Further equalities follow:
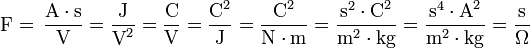
The most commonly used multiples and submultiples in electrical and electronic usage are the microfarad, nanofarad and picofarad.
Taken from Wikipedia










